 
This increasing sophistication of ideas over time should reassure teachers who find that, despite their best instructional efforts, their elementary students still do not fully understand these concepts.įinally, language and diagrams used in conversation and in textbooks can also lead to misunderstanding. Students also develop over the years a better understanding of the conservation of matter like water and air. As children age, they are better able to understand states of matter, particularly the more abstract concept of a gas. However, some of the misconceptions regarding states and changes of matter can actually be viewed as part of a developmental process. Numerous studies and anecdotal evidence show that students cling to these ideas even in the face of discrepant events and explicit instruction. Often, these ideas are formed well before a student arrives in science class – and serve their purpose well. In general, misconceptions result from students creating their own explanations for how the world works. Ice in all its forms (sea ice, glaciers, ice sheets, icebergs, permafrost) is part of the global water cycle. The water cycle only includes rain and snow. Water can evaporate from plants, animals, puddles, and the ground in addition to bodies of water. Water only gets evaporated from the ocean or lakes. 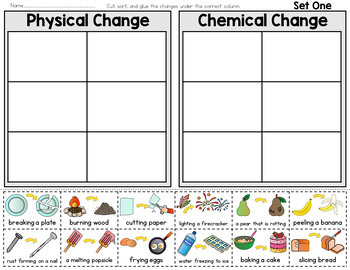
The water cycle involves evaporation of liquid water, condensation of water vapor, and precipitation (rain, sleet, hail, or snow). The water cycle involves freezing and melting of water. Additionally, students may understand the water cycle on a local level and not generalize the concept to a global scale or may not understand that water has been conserved throughout time. These concrete thinkers also have a difficult time with the idea of conservation of matter in terms of water vapor and air, making evaporation and condensation particularly difficult concepts. They see the water cycle primarily in terms of freezing and melting. Younger students tend to understand the water cycle by focusing on the properties of water. Matter expands when heated because the molecules are vibrating more quickly, loosening bonds, and increasing the space between adjacent atoms or molecules. Ice molecules have less kinetic energy than water molecules.Ĭondensation is when air turns into a liquid.Ĭondensation is water vapor in the air that cools enough to become a liquid.Ĭondensation on the outside of a container is water that seeped (or sweated) through the walls of the container.Ĭondensation of water vapor happens when the water vapor in air comes in contact with a cool surface.Įxpansion of matter is due to the expansion of the particles rather than increased space between the particles. Ice molecules are colder than water molecules. Water in an open container evaporates, changing from a liquid to a gas. Water in an open container is absorbed by the container, disappears, changes into air, or dries up and goes into the air. When water vapor condenses in the air it is visible as tiny water droplets. When steam is no longer visible it becomes air. When water boils and bubbles, the bubbles are air, oxygen or hydrogen, or heat.īubbles formed by boiling water consist of water vapor (steam). Otherwise, a chemical reaction has occurred and a chemical change is indicated.Misconceptions About States and Changes of Matter (Water) Students may think… 
Usually, if there is no color change, temperature change, precipitate formation, or gas production, the solution is a physical change. Solutions - Solutions are tricky because it may be hard to tell whether or not a chemical reaction has occurred when you mix together the materials.The reason alloying is not a chemical change is that the components retain their original identities. Alloys - Mixing together two or more metals is a physical change that is not reversible.Turning graphite into a diamond doesn't produce a chemical reaction. Crystallization - Crystallizing a solid does not produce a new molecule, even though the crystal will have different properties from other solids.Yet, you can separate the components of the mixture by allowing them to settle or by using a sieve. For example, if you mix together sand and water, you can pack the sand into a shape. Note the properties of a mixture may be different from its components. Mixtures - Mixing together materials where one is not soluble in the other is a physical change.This is a physical change because it's not permanent and no chemical reaction occurs. Magnetism - If you hold a magnet up to iron, you'll temporarily magnetize it.Phase Changes - Altering the temperature and/or pressure can change the phase of a material, yet its composition is unchanged,.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
